Monkeypox Virus IgM/IgG antibody rapid test kits
Monkeypox Virus IgM/IgG antibody rapid test kits,
monkeyox rash, Monkeypox diagnosis, Monkeypox Test, Monkeypox virus test monkeypox virus test kit monkeypox virus test price monkeypox virus test near me monkeypox virus pcr test monkeypox virus rapid test monkeypox virus lab test monkeypox virus antig,
Product details
Intended Use
The Monkeypox Virus IgM/IgG Antibody Rapid Test Kit is used for the qualitative detection of Monkeypox Virus IgM/IgG antibody in human serum, plasma or whole blood sample. It is intended for in vitro diagnostic use, and for professional use only.
Test Principle
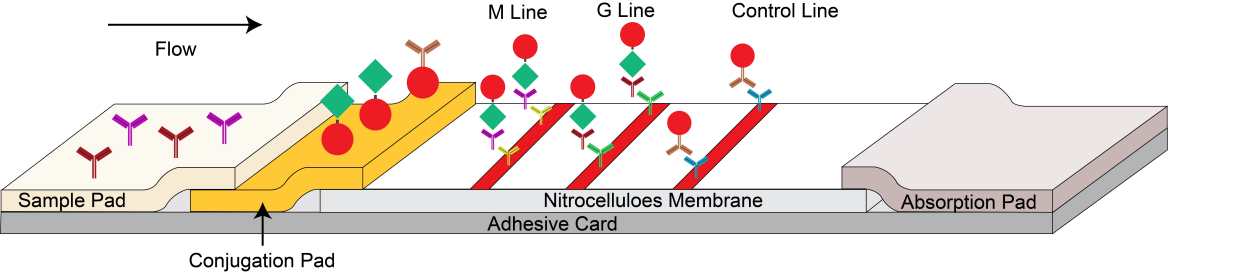
The Monkeypox Virus IgM/IgG test device has 3 pre-coated lines, “G” (Monkeypox IgG Test Line), “M” (Monkeypox IgM Test Line) and “C” (Control Line) on the surface of the membrane. The “Control Line” is used for procedural control. When a specimen is added to the sample well, anti-Monkeypox IgGs and IgMs in the specimen will react with recombinant Monkeypox virus envelope proteins conjugates and forms antibody -antigen complex. As the complex migrates along the test device via capillary action, it will be captured by the relevant anti-human IgG and or anti-human IgM immobilized in two test lines across the test device and generate a colored line. To serve as a procedural control, a colored line will always appear in the control line region, indicating that proper volume of specimen has been added and membrane wicking has occurred.
Main Contents
Components provided are listed in the table.
| Component REFREF | B030C-01 | B030C-05 | B030C-25 |
| Test Cassette | 1 test | 5 tests | 25 tests |
| Sample Diluent | 1 bottle | 5 bottles | 25 bottles |
| Disposable Lancet | 1 piece | 5 pcs | 25 pcs |
| Alcohol Pad | 1 piece | 5 pcs | 25 pcs |
| Disposable Dropper | 1 piece | 5 pcs | 25 pcs |
| Instructions For Use | 1 piece | 1 piece | 1 piece |
| Certificate of Conformity | 1 piece | 1 piece | 1 piece |
Operation Flow
- Step 1: Sampling
Collect human Serum/Plasma/Whole blood properly.
- Step 2: Testing
1. When ready to test, open the pouch at the notch and remove the device. Place
the test device on a clean, flat surface.
2. Fill the plastic dropper with the specimen. Holding the dropper vertically,
dispense 10µL of serum/plasma or 20µL of whole blood into the sample well,
making sure there are no air bubbles.
3. Immediately add 3 drops (about 100 µL) of sample diluent to sample well with
the bottle positioned vertically. Start counting.
- Step 3: Reading
15 mins later, read the results visually. (Note: do NOT read the results after 20 mins!)
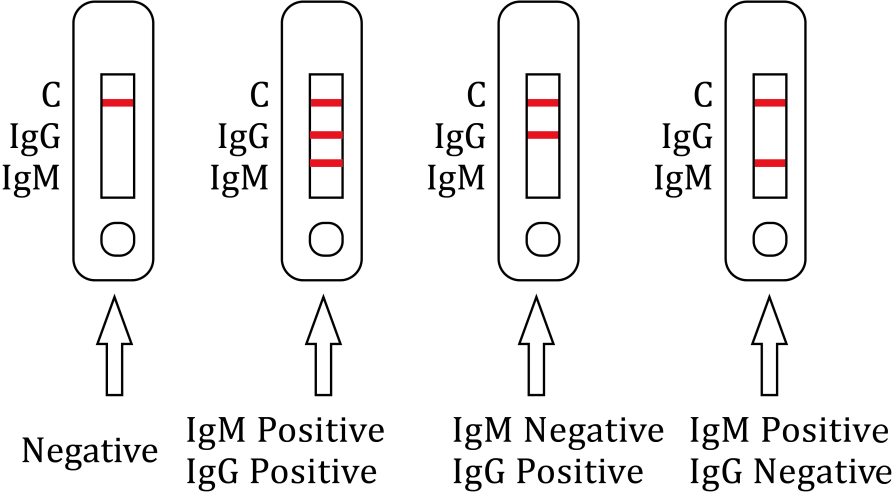
Result Interpretation
|
Positive |
Negative |
Invalid |
||
| -Positive IgM Result-
The control line (C) and IgM line (M) are visible on the test device. This is positive for IgM antibodies to monkeypox virus. |
-Positive IgG Result- The control line (C) and IgG line (G) are visible on the test device. This is positive for IgG antibodies to monkeypox virus. |
-Positive IgM & IgG-
The control line (C), IgM (M) and IgG line (G) are visible on the test device. This is positive for both IgM and IgG antibodies. |
Only the C line appears and the detection G line and M line do not appear. | No line appears in C line no matter G line and/or M line appear or not. |
Order Information
| Product Name | Cat. No | Size | Specimen | Shelf Life | Trans. & Sto. Temp. |
| Monkeypox Virus IgM/IgG Antibody Rapid Test Kit (LateralChromatography) | B030C-01 | 1 test/kit | S/P/WB | 24 Months | 2-30℃ |
| B030C-05 | 1 test/kit | ||||
| B009C-5 | 25 tests/kit |
Monkeypox virus test
Monkeypox is a viral disease that can be spread from infected animals to people. It primarily affects wild and domesticated non-human primates, but has also been known to infect humans. Monkeypox was first reported in 1958 in the Democratic Republic of Congo and was identified as a distinct clinical entity in humans in 1970 when it appeared in the United States.
This test is performed on any patient with symptoms of monkeypox infection, as well as on family members, close contacts, and others who have been exposed to a patient with monkeypox. The results are available within 24 hours.