OEM Malaria Test Malaria Test Medical Malaria Antigen Test Kit
abide by the contract”, conforms on the market requirement, joins within the market competition by its superior quality likewise as provides far more comprehensive and great company for shoppers to let them develop into huge winner. The pursue on the corporation, is definitely the clients’ gratification for OEM Malaria Test Malaria Test Medical Malaria Antigen Test Kit, It is our great honor to fulfill your needs.We sincerely hope we will cooperate along with you inside the close to foreseeable future.
abide by the contract”, conforms on the market requirement, joins within the market competition by its superior quality likewise as provides far more comprehensive and great company for shoppers to let them develop into huge winner. The pursue on the corporation, is definitely the clients’ gratification for China Malaria Rapid Test Kit and Malaria Test, Most problems between suppliers and clients are due to poor communication. Culturally, suppliers can be reluctant to question items they do not understand. We break down these barriers to ensure you get what you want to the level you expect, when you want it. Faster delivery time and the product you want is our Criterion .
Product details
Intended Use
Malaria antigen detection kit is designed as a simple, rapid, qualitative and cost effective method for the simultaneous detection and differentiation of Plasmodium falciparum (P.f) and Plasmodium vivax (P.v) in human whole blood or fingertip whole blood. This device is intended to be used as a screening test and used for the auxiliary diagnosis of P. f and P.v infection.
Test Principle
The Malaria antigen test kit (Lateral chromatography) is based on the principle of microsphere double antibody sandwich method to rapid qualitative determination of P.f/ P.v antigen in human whole blood or fingertip whole blood. The microsphere is marked in anti-HRP-2 antibody (specific to P.f) on the T1 band and anti-PLDH antibody(specific to P.v) on the T2 band, and anti-mouse IgG polyclonal antibody is coated on quality control area (C). When the sample contains malaria HRP2 or pLDH antigen and the concentration is higher than the minimum detection limit, which are allowed to react with the colloidal microsphere coated with Mal-antibody to form antibody-antigen complex. The complex then moves laterally on the membrane and respectively binds to the antibody immobilized on the membrane producing a pink line on the test region, which indicates a positive result. The presence of the control line demonstrates the test has been performed correctly regardless the presence of P.f/ P.v antigen.
Main Contents
Components provided are listed in the table.
| ComponentREF | B013C-01 | B013C-25 |
| Test Cassette | 1 test | 25 tests |
| Sample Diluent | 1 bottle | 1 bottle |
| Dropper | 1 piece | 25 PCS |
| Instructions For Use | 1 piece | 1 piece |
| Certificate of Conformity | 1 piece | 1 piece |
Operation Flow
Step 1: Sampling
Collect human whole blood or fingertip blood properly.
Step 2: Testing
1. Remove an extraction tube from the kit and a test box from the film bag by tearing the notch. Put it on the horizontal plane.
2. Open the inspection card aluminum foil bag. Remove the test card and place it horizontally on a table.
3. Add 60μL sample dilution solution immediately. Start counting.
Step 3: Reading
20 mins later, read the results visually. (Note: do NOT read the results after 30 minutes!)
Result Interpretation
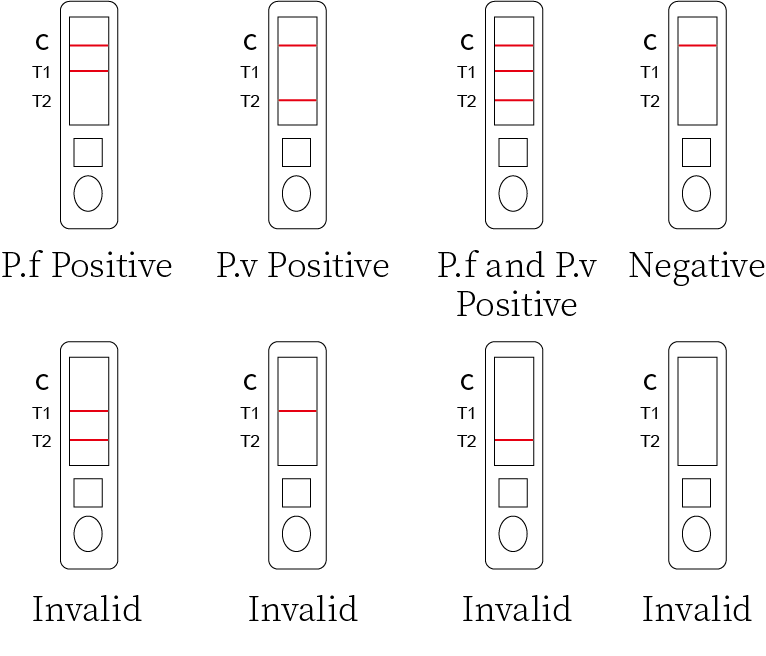
1.P.f Positive
The presence of two colored bands (“T1″ and “C”) within the result window indicates P.f Positive.
2.P.v Positive
The presence of two colored bands (“T2″and “C”) within the result window indicates P.v.
3.Positive. P.f and P.v Positive
The presence of three colored bands (“T1″,”T2″and “C”) within the result window may indicates mixed infection of P. f and Pan.
4.Negative Result
The presence of only control line(C) within the result window indicates a negative result.
5.Invalid Result
If no band appears in the control region(C), the test results are invalid regardless of the presence or absence of line in the test region(T). The direction may not have been followed correctly or the test may have deteriorated It is recommended that repeat the test using a new device.
Order Information
| Product Name | Cat. No | Size | Specimen | Shelf Life | Trans. & Sto. Temp. |
| Malaria HRP2/pLDH (P.f/P.v) Antigen Rapid Test Kit (Lateral chromatography) | B013C-01 | 1test/kit | Whole Blood/Fingertip Blood | 18 Months | 2-30℃ / 36-86℉ |
| B013C-25 | 25 tests/kit |
joins within the market competition by its superior quality likewise as provides far more comprehensive and great company for shoppers to let them develop into huge winner. The pursue on the corporation, is definitely the clients’ gratification for China OEM Malaria Test Malaria Test Medical Malaria P. F. /P. V. /Pan. Antigen/Antibody Test Kit, It is our great honor to fulfill your needs.We sincerely hope we will cooperate along with you inside the close to foreseeable future.
China OEM China Malaria Rapid Test Kit and Malaria Test, Most problems between suppliers and clients are due to poor communication. Culturally, suppliers can be reluctant to question items they do not understand. We break down these barriers to ensure you get what you want to the level you expect, when you want it. Faster delivery time and the product you want is our Criterion .